Institute of Science and Technology Austria (IST Austria), Klosterneuburg, Austria
Description
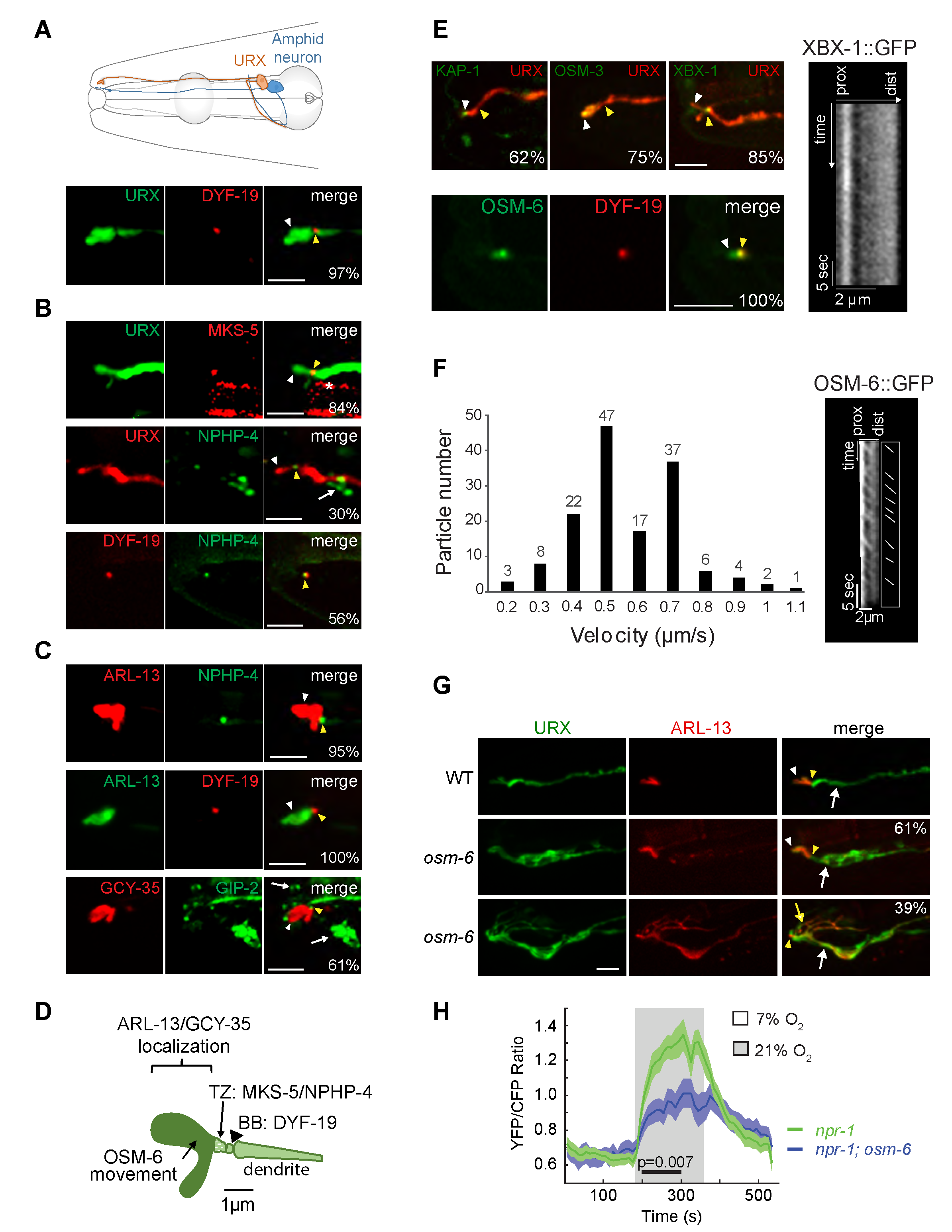
A subset of sensory neurons in C. elegans contains compartmentalized sensory structures termed cilia at their distal dendritic ends (Ward et al. 1975; Perkins et al. 1986; Doroquez et al. 2014). Cilia present on different sensory neuron types are specialized both in morphology and function, and are generated and maintained via shared and cell-specific molecules and mechanisms (Perkins et al. 1986; Evans et al. 2006; Mukhopadhyay et al. 2007; Mukhopadhyay et al. 2008; Morsci and Barr 2011; Doroquez et al. 2014; Silva et al. 2017). The bilaterally symmetric pair of URX oxygen-sensing neurons in the C. elegans head (Figure 1A) is thought to be non-ciliated (Ward et al. 1975; Doroquez et al. 2014) but nevertheless exhibits intriguing morphological similarities with ciliated sensory neurons. URX dendrites extend to the nose where they terminate in large bulb-like complex structures (Ward et al. 1975; Doroquez et al. 2014; Cebul et al. 2020) (Figure 1A). These structures concentrate oxygen-sensing signaling molecules (Gross et al. 2014; Mclachlan et al. 2018) suggesting that similar to cilia, these structures are specialized for sensory functions. Microtubule growth events similar to those observed in ciliated sensory neurons were also reported at the distal dendritic regions of URX, implying the presence of a microtubule organizer such as a remodeled basal body (Harterink et al. 2018). Moreover, a subset of ciliary genes is expressed in URX (Kunitomo et al. 2005; Harterink et al. 2018; Mclachlan et al. 2018). We tested the hypothesis that URX dendrites contain cilia at their distal ends.
We first examined the localization of proteins associated with different ciliary compartments in URX. The basal body component DYF-19/FBF1 (Wei et al. 2013) was enriched at single puncta at the distal dendritic ends of URX (Figure 1A). A similar localization pattern was previously reported for the centrosome-associated protein gamma-tubulin (Harterink et al. 2018). The MKS-5::tagRFP (Williams et al. 2011) and NPHP-4::GFP (Jauregui and Barr 2005; Williams et al. 2011) transition zone fusion proteins were also localized to single puncta at the URX dendritic tips, distal to the region of DYF-19 localization (Figure 1B). In addition, ARL-13::GFP, a well-characterized ciliary membrane marker (Cevik et al. 2010; Li et al. 2010) was restricted to a region distal to the DYF-19::tagRFP and NPHP-4::GFP puncta (Figure 1C). The mKate-tagged oxygen-sensing soluble guanylyl cyclase GCY-35 was also highly enriched at the distal dendritic ends of URX in a domain that was defined proximally by the GIP-2 component of gamma-TuRC expressed from its endogenous locus (Figure 1C) (Wang et al. 2015; Harterink et al. 2018). We noted that overexpression of ARL-13 and GCY-35 resulted in an expansion of the presumptive cilium-like structure in URX; overexpression of ARL-13 has previously been reported to alter cilia morphology in other cell types (Hori et al. 2008; Larkins et al. 2011). Together, these observations imply the presence of a cilium-like structure at the distal dendritic ends of URX that houses signaling molecules, and that is delineated proximally by a transition zone and basal body (summarized in Figure 1D). We speculate that the short length of this structure may have precluded its identification in previous ultrastructural studies (Ward et al. 1975; Perkins et al. 1986; Doroquez et al. 2014).
We next investigated whether intraflagellar transport (IFT) necessary for cilia generation and maintenance (Rosenbaum and Wittman, 2014) could be detected in this cilium-like organelle in URX. Although the KAP-1 kinesin-2 motor component, OSM-3 homodimeric motor, and XBX-1 dynein light chain subunit were localized to the very distal ends of the URX dendrites, we were unable to detect movement of these proteins under standard conditions (Figure 1E). Similar to the localization of these proteins, the IFT core component OSM-6::GFP was also present in a small domain distal to that occupied by the DYF-19::tagRFP puncta (Figure 1E). However, we observed anterograde movement of OSM-6::GFP in the URX dendritic ends at an average speed of 0.61 ± 0.20 µm/sec (Figure 1F), consistent with kinesin-2-mediated transport. We were unable to detect and/or quantify retrograde movement of OSM-6::GFP. Unlike in other characterized sensory cilia which typically exhibit robust IFT, episodes of OSM-6::GFP anterograde movement in the presumptive URX cilia-like structure were infrequent and could be observed in only ~20% of examined neurons under specific conditions (see Methods).
We asked whether IFT proteins are necessary for the structure and/or function of the URX cilium-like structure. Although we were unable to definitively determine whether the very short cilium-like structure was further truncated in osm-6 mutants, we found that ARL-13::tagRFP was mislocalized to the URX dendrite in all examined osm-6 mutants (Figure 1G). Interestingly, ~40% of URX neurons in osm-6 mutants also exhibited multiple branches emanating from their distal dendritic ends; these branches contained ARL-13::GFP (Figure 1G). Similar, albeit less extensive, branches (also referred to as ‘posterior projections’) have been reported from the distal ends of a subset of ciliated sensory neuron dendrites in animals mutant for IFT genes in C. elegans (Lewis and Hodgkin 1977; Perkins et al. 1986; Fujiwara et al. 1999; Murayama et al. 2005; Kunitomo and Iino 2008; Maurya et al. 2019).
URX responds to a 7%-21% rise in oxygen with a tonic increase in intracellular calcium levels (Zimmer et al. 2009; Busch et al. 2012). We found that only 45% (20/44) of URX neurons in osm-6(p811) mutants exhibited oxygen-evoked responses as assessed via changes in YC2.60 fluorescence, as compared to 100% of neurons (21/21) in wild-type animals. Moreover, response amplitudes were significantly decreased in responding URX neurons in osm-6 mutants (Figure 1H). We conclude that a subset of IFT molecules is necessary for the morphological and functional integrity of the cilia-like compartment in URX.
The sensory compartments at the distal ends of URX dendrites exhibit several features characteristic of cilia. Basal body, transition zone, IFT, and ciliary membrane and membrane-associated markers, including sensory signaling molecules, are localized in domains whose relative organization at the URX dendritic ends is similar to those present in bona fide sensory cilia. Moreover, OSM-6 appears to undergo IFT in a subset of URX cilia-like structures and is required for correct neuronal morphology, ciliary protein localization and sensory functions. However, we did not observe movement of any additional IFT proteins including motors. It is possible that motor movement in the short URX cilium is difficult to detect due to technical considerations. Alternatively, URX may employ cell-specific mechanisms including cell-specific motors to build its cilia. Finally, environmental manipulations have been shown to alter neuronal and cilia morphology including URX dendritic morphology in C. elegans (Albert and Riddle 1983; Mukhopadhyay et al. 2008; Procko et al. 2011; Schroeder et al. 2013; Cohn et al. 2019). Thus, trafficking in URX cilia may be modulated in a context-specific manner, perhaps to regulate cilium length or protein composition in response to specific cues. In the future, it will be interesting to correlate IFT in URX cilia with neuronal responses under defined external and internal conditions.
Methods
Request a detailed protocolC. elegans growth
C. elegans strains were maintained at 20°C under normoxic conditions on standard NGM agar plates seeded with E. coli OP50 unless specified otherwise (all strains used in this work are indicated in Table 1). Mutant and transgenic strains were generated using standard methods. The presence of the desired mutation was confirmed by sequencing. Plasmids were injected at 1-10 ng/µl (see Table 2) with co-injection markers (unc-122p::gfp, unc-122p::rfp or unc-122p::dsRed) injected at 30 ng/µl.
Molecular biology
All DNA constructs were generated using standard cloning techniques, subcloned into the pPD95.77 C. elegans expression plasmid (A.Fire, Stanford University) and validated by restriction enzyme digestion and sequencing. Plasmids used in this work are indicated in Table 2.
Microscopy
One day-old adults were anesthetized using 10mM tetramisole (Sigma), diluted in M9 buffer and mounted onto 10% agarose pads. Live anesthetized animals were imaged on an inverted spinning disk microscope (Zeiss Axio Observer with a Yokogawa CSU-22 spinning disk confocal head). Images of protein localization were generated by collecting optical sections (z-stack) every 0.2-0.27 µm using Plan Apochromat 100x/1.40 NA or 63X/1.20 NA oil immersion objectives. Maximum intensity projection (z-stacks) images were generated using SlideBook 6.0 software (Intelligent Imaging Innovations, 3i). Enhancement of brightness and contrast across the entire image, as well as image rotations were performed using ImageJ/Fiji (NIH) software. Each strain was imaged independently on at least two different days. Two or more transgenic lines were generated and examined for each transgenic strain.
IFT
L4 animals were picked 18-24 hours prior to imaging and maintained at 15°C on unseeded NGM plates under normoxic conditions. OSM-6 IFT movement was observed only under these conditions in very young adults. 2D timelapse videos of OSM-6 and XBX-1 were recorded using SlideBook 6.0 software (Intelligent Imaging Innovations, 3i) using an inverted spinning disk confocal microscope (Zeiss Axio Observer with a Yokogawa CSU-22 spinning disk confocal head) using a Plan Apochromat 63X NA 1.2 oil immersion objective. The kymographs were generated using the MultipleKymographs plugin in ImageJ/Fiji (NIH). Individual particle tracks were manually traced.
Calcium imaging
Imaging of the calcium response in URX neurons was carried out across 3 days in animals expressing gcy-37p::YC2.60 as previously described (Chen et al. 2017). Briefly, movies were recorded with a Nikon AZ100 microscope equipped with a Nikon ×2 AZ-Plan Fluor objective and Hamamatsu ORCA-FLASH4.0 cameras using NIS-Elements software and 500ms exposure time. Excitation light produced by a Nikon Intensilight C-HGFI was passed through a 438/24nm filter and a Semrock FF458DiO2 dichroic. The emission light was split using a Cairn Research TwinCam dual camera adapter and passed through CFP and YFP filters (483/32nm and 542/27nm respectively), and a DC/T510LPXRXTUf2 dichroic. Dermabond adhesive was used to immobilize young adults on agar pads while keeping the nose exposed. 1ml of concentrated OP50 in M9 buffer was applied to the head and allowed to dry before the slide was transferred to the imaging chamber. In the imaging chamber, animals were exposed to 7% oxygen for 2 minutes before starting the experiment in which 7% then 21% then 7% oxygen concentrations were sequentially flowed into the imaging chamber for 3 min intervals. The recorded data was processed using Neuron Analyzer, a custom-written Matlab program.
Reagents
Table 1. List of strains used in this work.
| Strain | Genotype | Source |
| PY11327 | iaIs25 [gcy-37p::gfp, unc-119(+)]; oyEx643 [gcy-36p::dyf-19::tagRfp, unc-122p::mCherry] | Inna Nechipurenko |
| PY10377-79 | Ex[gcy-32p::gfp, gcy-32p::mks-5::tagRfp; unc-122p::Rfp] Lines 2A, 6A, 21A | This work |
| PY10380 | Ex[nphp-4p::nphp-4::gfp, gcy-36p::tagRfp, unc-122p::Rfp] Line 2A | This work |
| PY10381 | Ex[gcy-32p::osm-3b::gfp, gcy-36p::tagRfp, unc-122p::Rfp] Line 10B | This work |
| PY10382-84 | Ex[gcy-32p::kap-1::gfp, gcy-36p::tagRfp, unc-122p::Rfp] Lines 7E, 11B, 16C | This work |
| PY11316 | iaIs25 [gcy-37p::gfp, unc-119(+)]; oyEx645 [gcy-32p::arl-13::tagRfp, unc-122p::mCherry] | Inna Nechipurenko |
| PY10386 | osm-6(p811); iaIs25 [gcy-37p::gfp]; Ex[gcy-32p::arl-13::tagRfp, unc-122p::dsRed] | This work |
| PY10357 | Ex[gcy-32p::osm-6::gfp, gcy-36p::tagRfp, unc-122p::Rfp] | This work |
| PY10388-90 | Ex[gcy-36p::xbx-1::gfp, gcy-36p::tagRfp, unc-122p::Rfp] Lines 13C, 13A, 4A | This work |
| PY10391-94 | Ex[gcy-32p::arl-13::tagRfp, gcy-36p::nphp-4::gfp, unc-122p::gfp] Lines 1-4 | This work |
| PY10395 | Ex[gcy-36p::nphp-4::gfp, gcy-36p::dyf-19::tagRfp, unc-122p::gfp] Line 8 | This work |
| PY10396-99 | Ex[gcy-36p::dyf-19::tagRfp; gcy-32p::osm-6::gfp, unc-122p::Rfp] Lines 2, 4, 5A, 5B | This work |
| PY10310 | gip-2(lt19[gip-2::gfp::loxP::cb-unc-119(+)::loxP]); hrtSi57[gcy-36p::gcy-35::mKate-intra] | Martin Harterink |
| PY10387 | Ex[gcy-36p::dyf-19::tagRfp, gcy32p::arl-13::gfp, unc-122p::Rfp] Line 2 | This work |
| PY10352 | osm-6(p811); npr-1 (ad609), Ex[gcy-37p::YC2.60] | This work |
Table 2. List of plasmids used in this work.
| Plasmid | Description | Source (injection concentration) |
| PSAB1231 | gcy-32p::mks-5cDNA::tagRfp | Inna Nechipurenko (5 ng/µl) |
| PSAB1224 | gcy-36p::tagRfp | This work (5 ng/µl) |
| PSAB1028 | nphp-4p::nphp-4::gfp | Maureen Barr (10 ng/µl) |
| PSAB1225 | gcy-32p::osm-3b::gfp | This work (10 ng/µl) |
| PSAB1226 | gcy-32p::kap-1::gfp | This work (10 ng/µl) |
| PSAB1215 | gcy-32p::arl-13::tagRfp | Inna Nechipurenko (10 ng/µl) |
| PSAB1227 | gcy-32p::osm-6::gfp | This work (10 ng/µl) |
| PSAB1228 | gcy-36p::xbx-1::gfp | This work (10 ng/µl) |
| PSAB1229 | gcy-36p::nphp-4::gfp | This work (10 ng/µl or 1ng/µl) |
| PSAB1216 | gcy-36p::dyf-19::tagRfp | Inna Nechipurenko (1 ng/µl or 5 ng/µl) |
| PSAB1230 | gcy-32p::arl-13::gfp | This work (10 ng/µl) |
Acknowledgments
We thank Maureen Barr, Martin Harterink, Max Heiman and Inna Nechipurenko for reagents, the Caenorhabditis Genetics Center for strains, and the Sengupta lab for comments and advice.
References
Funding
This work was funded in part by the NIH (R35 GM122463 – P.S., and F32 DC018453 – A.P.), and the EMBO (ALTF 302-2019 – N.A-W.).
Reviewed By
Oliver BlacqueHistory
Received: August 10, 2020Revision received: September 8, 2020
Accepted: September 9, 2020
Published: September 20, 2020
Copyright
© 2020 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Citation
Kazatskaya, A; Yuan, L; Amin-Wetzel, N; Philbrook, A; de Bono, M; Sengupta, P (2020). The URX oxygen-sensing neurons in C. elegans are ciliated. microPublication Biology. 10.17912/micropub.biology.000303.Download: RIS BibTeX