Division of Aging Biology, National Institute on Aging, Bethesda, Maryland 20892, USA
Department of Molecular Biology and Biochemistry, Rutgers University, Piscataway, New Jersey 08854, USA
The Buck Institute for Research on Aging, Novato, California 94945, USA
Description
The Caenorhabditis Intervention Testing Program (CITP) is a multi-institutional, National Institutes of Aging (NIA)-funded consortium charged with identifying chemical compounds that robustly extend lifespan in a genetically diverse panel of Caenorhabditis strains. Compounds are prioritized for screening if they are highly ranked via computational prediction for lifespan or healthspan effects (Coleman-Hulbert et al. 2019), if they are predicted to engage known lifespan regulating pathways, or if they have previously been reported as extending lifespan or healthspan in model systems (Lucanic et al. 2017). β-guanidinopropionic acid (β-GPA) is a creatine analog (Shields and Whitehair 1973), commonly used as a dietary supplement, and has been shown to extend lifespan in Drosophila under stress via 5′ AMP-activated protein kinase (AMPK) activity (Yang et al. 2015). The AMPK pathway is conserved in nematodes and humans (Apfeld et al. 2004) and is involved in multiple pathways affecting stress response and metabolism (Wang et al. 2012).
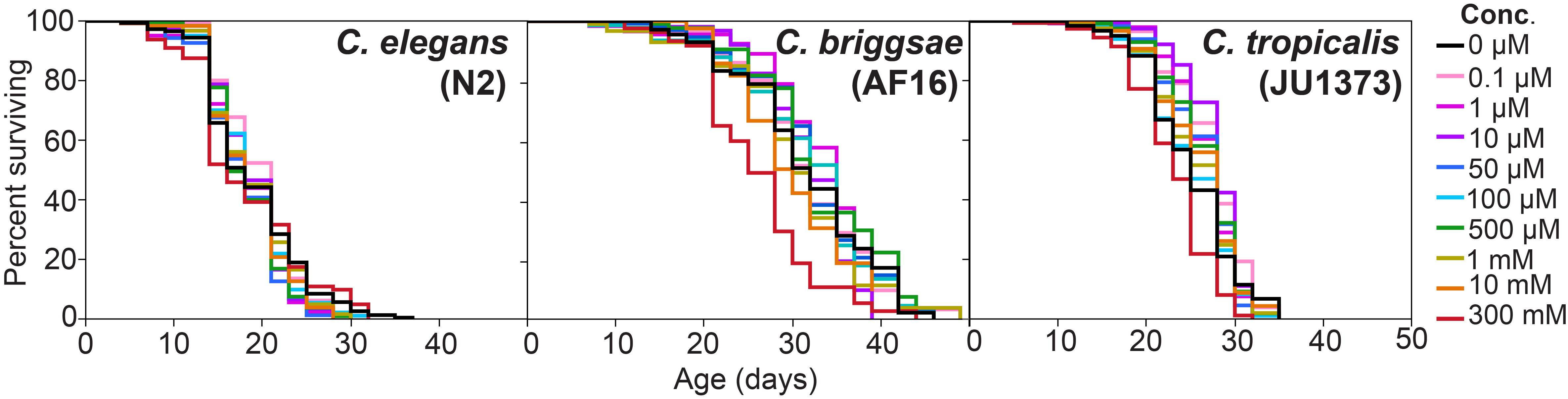
We assayed lifespan in response to β-GPA exposure in three Caenorhabditis species using our previously published workflow (Lucanic et al. 2017). In brief, worms were age-synchronized by timed egg-lays on standard 60 mm diameter Nematode Growth Media (NGM) plates and transferred at a density of 50 individuals per 35 mm treated plate in triplicate when they reached adulthood (for control plates, there were six replicates of 50 animals each). β-GPA (Sigma-Aldrich) was dissolved in water and diluted appropriately such that addition of 125 µl of solution to 35 mm diameter plates containing NGM with lawns of E. coli OP50-1 and 51 µm FUdR would generate the following final β-GPA concentrations: 0.1 µM, 1 µM, 10 µM, 50 µM, 100 µM, 500 µM, 1 mM, 10 mM and 300 mM. Worms were maintained at 20 °C and moved to fresh plates on the first, second, and fourth (C. tropicalis) or fifth (C. elegans and C. briggsae) day of adulthood, then once weekly afterward. Due to our previous experience with compound interventions that potentially alter bacterial viability through transient pH changes upon plate treatment (Banse et al. 2019), we investigated the effects of β-GPA on our assay plates. At the above listed concentrations, β-GPA-treated plates had a pH of 6.5 and the bacterial lawns survived treatment when tested by replica plating. Thrice weekly, we observed animals for spontaneous movement or movement after gentle perturbation with a 0.2 mm diameter platinum wire. Death was scored as a lack of movement.
Our results indicate that β-GPA does not extend lifespan in the three nematode species at the concentrations tested here; in fact, in only one instance was an effect detected and, in that case, the compound reduced lifespan (Fig. 1). This conclusion is based on one biological replicate per concentration and, as such, could be considered preliminary. While interventions may be ineffective due to a range of causes, including permeability barriers, compound stability in vivo, and metabolism by the bacterial food source, we believe that the lack of response in this study was due to a lack of physiological relevance in C. elegans. In Drosophila and mammalian models, β-GPA reduces the level of intracellular phosphocreatine that can be used by creatine kinases to regenerate ATP (Oudman et al. 2013; Yang et al. 2015) resulting in a decreased cellular ATP/AMP ratio, which activates AMPK and ultimately increases lifespan and stress-resistance. Our a priori expectation for lifespan extension in C. elegans was built on the observations that: (1) creatine is reported as detectible in C. elegans (Atherton et al. 2008; Jones et al. 2012; Wan et al. 2017); (2) C. elegans has a creatine-like kinase, ARGK-1, whose activity modulates AMPK signaling (McQuary et al. 2016); (3) modulation of AMPK (Apfeld et al. 2004; Greer et al. 2007) and ARGK-1 (McQuary et al. 2016) in C. elegans can affect lifespan and stress resistance; and (4) regulation of lifespan by insulin signaling is partially dependent on AMPK signaling in C. elegans (Tullet et al. 2014). As such, we were surprised to find no changes in lifespan upon treatment with a creatine analog. One possible explanation is that, despite the similarity between ARGK-1 and mammalian creatine kinase, the enzymes’ substrates differ. Biochemical analysis suggests that ARGK-1 uses arginine instead of creatine as a substrate to recharge ADP (Fraga et al. 2015). Additionally, it has been postulated that the biochemical characterization of C. elegans metabolites may have misidentified creatine, and that creatine is not relevant to C. elegans physiology (Witting et al. 2018). Given these caveats, it may not be surprising that β-GPA does not alter Caenorhabditis lifespan.
Acknowledgments
We acknowledge all of the members of the Lithgow, Driscoll and Phillips labs for helpful discussions. We thank the CITP Advisory Committee and Ronald Kohanski (National Institute on Aging) for extensive discussion. Strains were provided by the CGC, which is funded by NIH Office of Research Infrastructure Programs (P40 OD010440).
References
Funding
This work was supported by National Institutes of Health grants U01 AG045864 to MD, UO1 AG045844 to GJL, and U01 AG045829 and U24 AG056052 to PCP.
Reviewed By
AnonymousHistory
Received: December 20, 2019Accepted: December 29, 2019
Published: January 2, 2020
Copyright
© 2020 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Citation
Coleman-Hulbert, AL; Johnson, E; Sedore, CA; Banse, SA; Guo, M; Driscoll, M; Lithgow, GJ; Phillips, PC (2020). Caenorhabditis Intervention Testing Program: the creatine analog β-guanidinopropionic acid does not extend lifespan in nematodes. microPublication Biology. 10.17912/micropub.biology.000207.Download: RIS BibTeX