Description
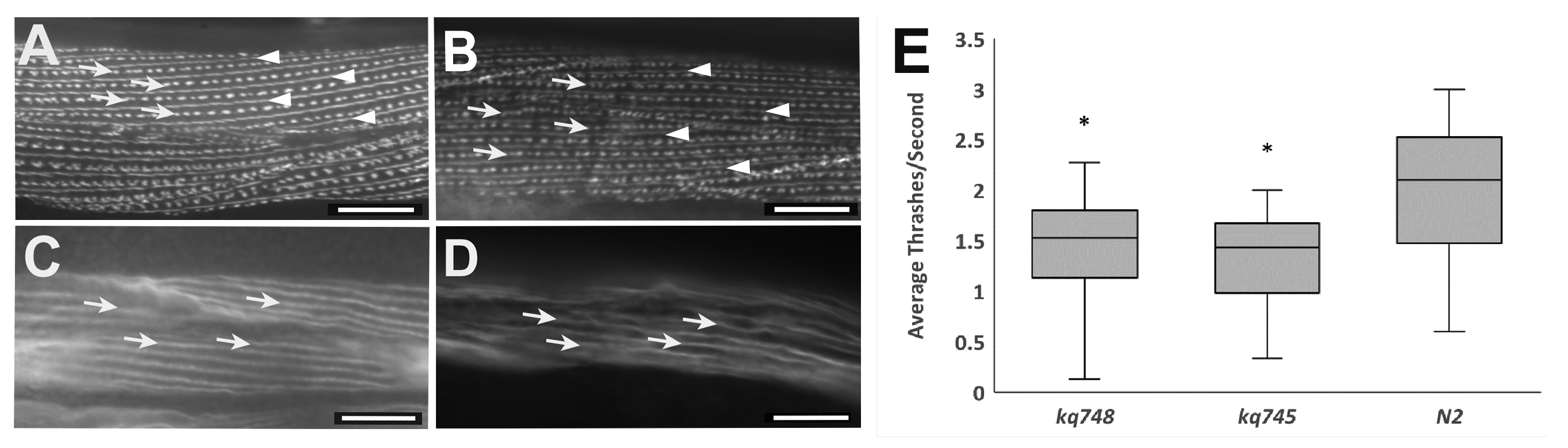
The unc-52 gene in Caenorhabditis elegans encodes for the protein UNC-52 and is a homolog of the mammalian gene Heparan Sulfate Proteoglycan 2 (HSPG2), which encodes for the protein perlecan (Rogalski et al., 1993; Mullen et al., 1999). HSPG2 is implicated in the human diseases Schwartz-Jampel Syndrome type 1 and Dyssegmental Dysplasia, Silverman-Handmaker type (Arikawa-Hirasawa et al., 2001; Stum et al., 2006). The mutated unc-52 gene expresses the phenotype for uncoordinated movement (“Unc”), which involves progressive paralysis and retarded sarcomere construction (Martinez et al., 2018). The UNC-52 protein is localized in the striated muscle dense bodies and the basement membrane, where it plays an important role in developmental processes such as cell adhesion, cell migration, and signal transduction (Kihira et al., 2012). Within the unc-52 gene, an RGD (Arg- Gly- Asp) sequence is located at amino acid locations 746, 747, and 748 in exon 7. Exon 7, containing RGD748, is included in all three major isoforms, short (S), medium (M), and long (L) (Mullen et al., 1999). The RGD sequence is part of the Laminin IV type A domain and primarily functions as a cell attachment and adhesion site for integrins (Rogalski et al., 1993; Mullen et al., 1999). In this study, two separate mutations were performed on this unc-52 RGD sequence using CRISPR-Cas9 technology. The unc-52(kq748) mutation replaced the aspartic acid (D) located at the 748 amino acid position with a glutamic acid (E) (Takahashi et al., 2007). The unc-52(kq745) mutation removed the RGD sequence by deleting amino acids 746, 747, and 748. Previous studies have shown that unc-52 gene mutations cause the disorganized distribution of pat-3 β integrin, which is a receptor for extracellular matrix proteins (Rogalski et al., 1995). In order to study the cellular phenotypes of the unc-52(kq748) mutation, a double mutant was created: pat-3::GFP; unc-52 (kq748). Staining showed that localization of the pat-3::GFP reporter in the double RGD mutant appeared normal, with dense bodies and M-lines alternating along the muscle filaments (Figures 1A and 1B). In order to visualize the actin filaments in the body wall muscles of N2 wild type and unc-52(kq748) mutants, staining was performed using 0.4 U/mL rhodamine-conjugated phalloidin, Thermo Fisher Scientific, Waltham, MA (Figures 1C and 1D). This unc-52(kq748) staining showed no obvious abnormalities outside of interruptions due to fixation and slicing procedures (Figure 1D). Additional assays were performed to investigate any other physical or behavioral phenotypes. The thrashing assay, in which worms were placed in drops of M9 buffer and the number of thrashes were counted for 15 seconds, showed a 27% average decrease in thrashes per second for the unc-52(kq748) mutants compared to the N2 worms, and a 33% decrease in unc-52(kq745) mutants (Figure 1E). A one-way ANOVA statistical test was performed to check the statistical significance of these results. The p-value from the ANOVA test was 3.1×10-9, which is less than the 0.05 needed to be statistically significant. Post hoc testing was then done to see which groups differed. Both the unc-52(kq748) and unc-52(kq745) groups differed significantly from the N2 wild-type group, as shown by p-values of 7.1×10-6 and 4.63×10-8 respectively. These results show that there was a statistically significant decrease in motility for both unc-52(kq748) and unc-52(kq745) mutants.
Methods
Request a detailed protocolCRISPR-Cas9 technology was used to create both the unc-52(kq748) and unc-52(kq745) mutations. For the unc-52(kq748) mutation, the DNA repair template (UNC52RGE748), crRNA (UNC52RGE748), tracrRNA (cat. #1072532), Cas9 nuclease (cat. #1081058), and a dpy-10 crRNA co-CRISPR marker were mixed and micro-injected into wild-type N2 worms, which served as the parent (P0) generation. F1 worms that carried the mutation were then identified and isolated using the dpy-10 marker (Arribere et al., 2014; Paix et al., 2015) and underwent PCR genotyping using mutant specific primers (UNC52RGE748SEQF and UNC52RGE748SEQR) to screen for the RGE mutation. The F2 generation then also underwent PCR screening using both a mutant specific primer (UNC52RGE748SEQF) and a wild-type specific primer (UNC52RGD748WTF) to isolate non-dumpy homozygous mutants. These homozygous mutant PCR products were sequenced to validate the success of the mutation (Psomagen Inc, Rockville, MD). This process was repeated for the unc-52(kq745) mutation using the DNA repair template (UNC52RGD748D) and the primers (UNC52RGD748DF and UNC52RGDWTF). The DNA repair template and all other oligos were designed at IDT Inc., Coralville, IA. A double mutant was then created by crossing unc-52(kq748) II males with NK358 pat-3::GFP III hermaphrodites. Homozygous double mutants from the F2 generation were identified as green-fluorescent progeny and confirmed through PCR genotyping. Staining was performed by collecting mutants and fixing them with ice-cold methanol for 5 minutes. Images were captured using a Nikon Eclipse Ni-U epifluorescence microscope and processed with NIS Elements (version 5.02). Once the homozygous mutants were identified and isolated, phenotypic assays were conducted on 50 young adult worms from both homozygous mutants and N2 wild-type worms in order to characterize the phenotypes for both the unc-52(kq748) and unc-52(kq745) mutations. A one-way ANOVA test was then run to confirm statistical significance of the results. Since the p-value for the ANOVA test was less than 0.05, post hoc tests were run to determine which groups differed (JMP, version 15, Cary, NC).
crRNA sequences (5’ to 3’)
UNC52RGD748 TTTCTATTGATTATGCTCGT
(dpy-10) ZQDP10A GCUACCAUAGGCACCACGAG
Repair templates (5’ to 3’)
| UNC52RGE748 | caaaaatatgtgttcaaccatcactttcttcagGTGTCAATCGACTACGCAAGAGGCGAACGTGACCAGCTCGAGCTCACCACCTCGGACTCCCG |
| UNC52RGD748D | caaaaatatgtgttcaaccatcactttcttcagGTGTCAATCGACTACGCAAGAGATCAACTCGAGCTCACCACCTCGGACTCCCGCCAACCATA |
Primers
UNC52RGE748SEQF TTCCTTGCTTCTGCTCAGGT
UNC52RGE748SEQR TGATCGGAGTTGCCATTTCCA
UNC52RGD748WTF TTCTATTGATTATGCTCGTGGGGAT
UNC52RGE748F TCAATCGACTACGCAAGAGGC
UNC52RGD748DF GTCAATCGACTACGCAAGAGATCAA
Reagents
BU748 unc-52(kq748) II, BU745 unc-52(kq745) II, and BU111 unc-52(kq748); pat-3::GFP are available upon request. NK358 pat-3::GFP III was purchased from Caenorhabditis Genetics Center, Minneapolis, MN.
Acknowledgments
These mutant lines were created during the course of BIO 4108 Cell and Developmental Biology Lab at Baylor University. Wild-type Bristol strain, N2, and fluorescence strains were purchased from the Caenorhabditis Genetics Center at the University of Minnesota, Minneapolis, MN.
References
Funding
Funding for BIO 4108 is provided by Baylor University.
Reviewed By
Don MoermanHistory
Received: May 4, 2020Revision received: May 9, 2020
Accepted: May 10, 2020
Published: May 17, 2020
Copyright
© 2020 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Citation
Wilsey, R; Hodge, S; Kenney, K; Wahl, J; Jaffery, R; Brau, A; Qiu, Z; Lee, M (2020). Two alleles of unc-52 locus disrupting potential cell-binding motif of UNC-52. microPublication Biology. 10.17912/micropub.biology.000250.Download: RIS BibTeX