University of Sydney
Description
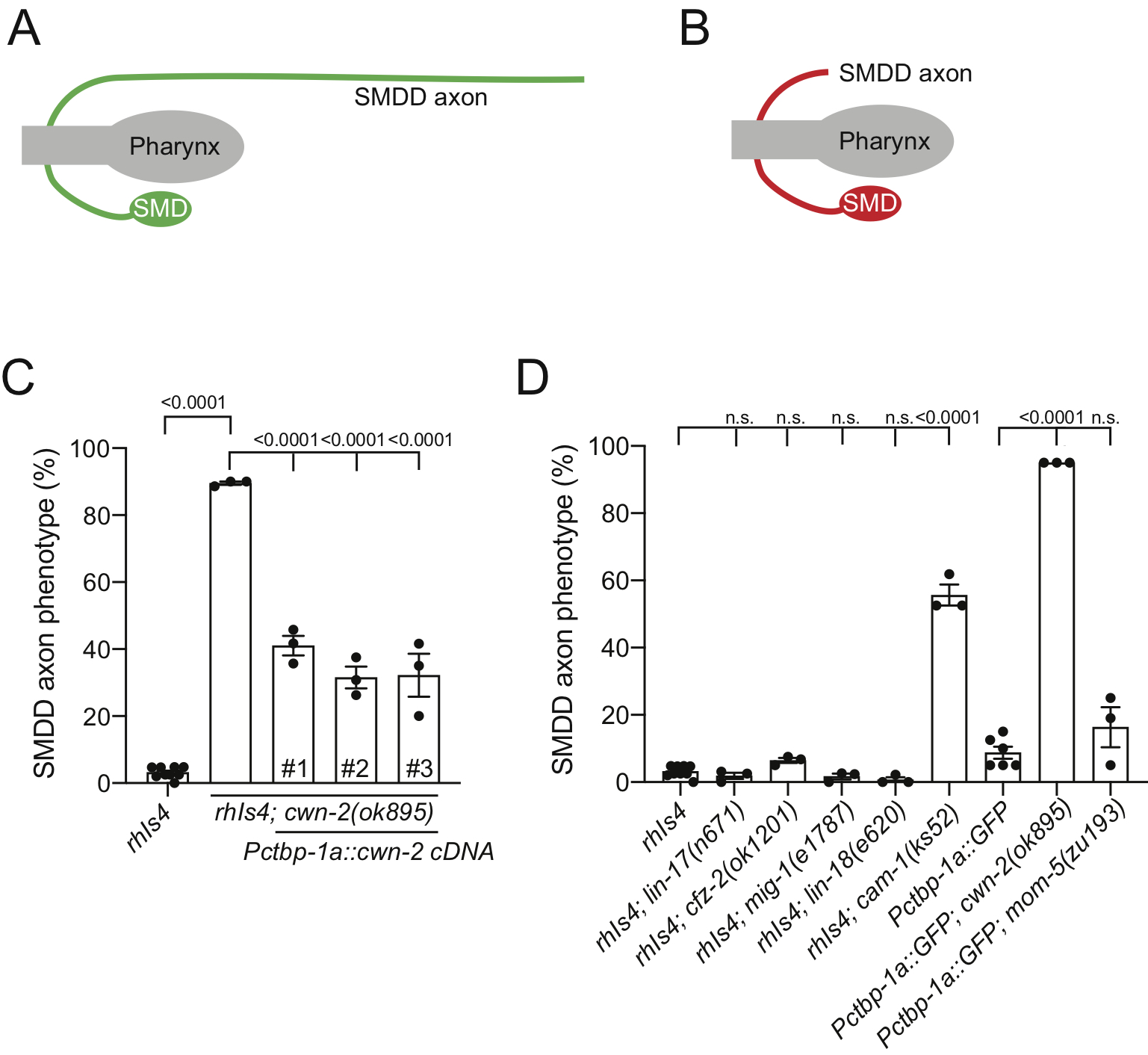
CWN-2 is one of five C. elegans Wnt ligands, with known roles in neuron migration, axon guidance and nerve ring placement (reviewed in (Sawa and Korswagen, 2013)). CWN-2 is highly expressed in the SMDD neurons (Kennerdell et al., 2009; Taylor et al., 2019), and therefore we were interested in determining whether CWN-2 regulates SMDD axonal development.
We used the cwn-2(ok895) deletion allele to analyze SMDD axonal development. We found that loss of cwn-2 caused a defective SMDD axonal phenotype, where axons do not extend along the dorsal sublateral cord, suggesting that the axons did not exit the nerve ring due to defects in axon outgrowth or guidance (Figure 1A-C). To ensure that this phenotype was due to the loss of CWN-2 function, we transgenically expressed cwn-2 cDNA driven by the ctbp-1a promoter, which drives expression in SMDD and ten other head neurons (Sherry et al., 2020). Expressing cwn-2 using this promoter partially rescues the defective axonal phenotype (Figure 1C). This suggests that CWN-2 controls SMDD development in an autocrine manner and/or non-autonomously from other head neurons.
Next, we investigated which Wnt receptor controls SMDD axonal development. There are six Wnt receptors encoded in the C. elegans genome: four Frizzled receptors (LIN-17, CFZ-2, MIG-1 and MOM-5,), one Ror receptor (CAM-1) and one Ryk receptor (LIN-18) (Sawa and Korswagen, 2013). We analyzed the effect of loss-of-function mutations for each receptor and found that loss of cam-1, but not the other receptors, caused defective SMDD axonal development (Figure 1D). CAM-1 was previously shown to function as a CWN-2 receptor for SIA, SIB and RMED/V posterior-directed neurite outgrowth (Kennerdell et al., 2009; Song et al., 2010; Wang and Ding, 2018). The SIA, SIB, RMED/V and SMDD motor neuron cell bodies are all positioned in the nerve ring and extend posteriorly-directed axons (White et al., 1986). Therefore, our data show that CWN-2 and CAM-1 have a common role in controlling axon extension in posteriorly-directed lateral or sublateral axons.
Methods
Request a detailed protocolC. elegans strains were grown using standard growth conditions on NGM agar at 20 °C and fed with Escherichia coli OP50. Neuroanatomical reporter strains used – rhIs4 [Pglr-1::GFP] and rpEx1739[Pctbp-1a::GFP]. Homozygous mom-5 mutant animals were selected based on the Unc phenotype caused by the linked unc-13(e1091) mutation. Day two adult hermaphrodites were anesthetized with 0.2% levamisole hydrochloride on 5% agarose pads for anatomical scoring. Schematics of axonal defects are shown instead of images as bright GFP expression in multiple cell bodies around the pharynx obscure imaging of axon defects. Refer to (Sherry et al., 2020) for images of the SMDD axons posterior to the pharynx. ‘Defective SMDD axonal phenotype (%)’ indicates the percentage of SMDD axons that do not extend along the dorsal sublateral cord. Two SMDD axons (L and R) were scored per animal. For cwn-2(ok895) rescue experiments, transgenic animals were compared to the cwn-2(ok895) mutant. Three biological replicates were performed, and statistical significance was assessed by one-way ANOVA followed by Tukey’s multiple comparisons tests.
Cloning was performed using Takara In-Fusion® restriction-free cloning. The Pctbp-1a::cwn-2 rescue construct was generated by cloning the cwn-2 1083 bp cDNA sequence, amplified from C. elegans cDNA, into a Pctbp-1a::GFP vector to replace the GFP sequence (Sherry et al., 2020). Rescue constructs were injected into rhIs4; cwn-2(ok895) mutant backgrounds at 2 ng/μl with Punc-122::GFP (20 ng/μl) as injection marker.
Reagents
Strains
HRN169 rhIs4 [Pglr-1::GFP] III
HRN498 rhIs4 III; cwn-2(ok895) IV
RJP4553 rhIs4 III; cwn-2(ok895) IV; rpEx2027 [Pctbp-1a::cwn-2] line #1
RJP4554 rhIs4 III; cwn-2(ok895) IV; rpEx2028 [Pctbp-1a::cwn-2] line #2
RJP4555 rhIs4 III; cwn-2(ok895) IV; rpEx2029 [Pctbp-1a::cwn-2] line #3
RJP4485 mig-1(e1787) I; rhIs4 III
RJP4486 lin-17(n671) I; rhIs4 III
RJP4534 rhIs4 III; cfz-2(ok1201) V
RJP4688 cam-1(ks52) II; rhIs4 III
RJP4715 rhIs4 III; lin-18(e620) X
RJP4076 rpEx1739 [Pctbp-1a::GFP]
RJP4598 cwn-2(ok895) IV; rpEx1739 [Pctbp-1a::GFP]
RJP4624 mom-5(zu193) unc-13(e1091)/hT2 III; rpEx1739 [Pctbp-1a::GFP]
Acknowledgments
We thank Cori Bargmann for the lin-18(e620) allele and the Caenorhabditis Genetics Center (University of Minnesota), which is funded by NIH Office of Research Infrastructure Programs (P40 OD010440), for strains.
References
Funding
This work was supported by an Australian Government Research Training Program (RTP) Scholarship to T.S, and the National Health and Medical Research Council (GNT1137645 to R.P).
Reviewed By
AnonymousHistory
Received: November 3, 2020Revision received: November 16, 2020
Accepted: November 23, 2020
Published: November 25, 2020
Copyright
© 2020 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Citation
Sherry, T; Nicholas, HR; Pocock, R (2020). CWN-2/Wnt regulates SMDD axonal development. microPublication Biology. 10.17912/micropub.biology.000337.Download: RIS BibTeX